

Treating the Female Patient:
The Female Menstrual CycleThis section was compiled by Frank M. Painter, D.C.
Send all comments or additions to: Frankp@chiro.org




Thanks to Dr. Diane Benizzi DiMarco and New York Chiropractic College for access to this article!
Premenstrual syndrome, Premenstrual Dysphoric Disorder and Primary Dysmenorrhea account for as much as 30 billion dollars of lost wages per year. There are more than 150 symptoms that contribute to the syndrome of PMS, PMDD and primary dysmenorrhea. [1, 2] With this in mind, it is safe to assume that most of society has been exposed to the affects of pre-menses in some way, either directly, through a relation, spouse or friend. The information contained is intended to provide the clinician with a viable alternative to invasive management in treating PMS, PMDD, and Dysmenorrhea. The neuromuscular system provides an excellent source to controlling the myriad symptoms associated with pre-menses.
Understanding the Female Cycle
The female reproductive system is cyclic in nature occurring approximately every 28 days. The cycle is broken down starting from day one to day twenty-eight. Day one of the cycle is the start of menstrual bleeding which continues through to day five. Day five through day fourteen is called the proliferation phase. Day thirteen is considered the ovarian phase, and days fourteen to twenty eight are known as the luteal or secretory phase. The female menstrual cycle which is earmarked by its’ regular, periodic hormonal fluctuations is largely influenced by hormones such as estrogen, progesterone, and leutenizing hormone, which are the most widely discussed and known biochemicals influencing female reproduction. Each hormone is secreted in a timely fashion and works in synthesis with each other. The first day of the menstrual cycle is identified by menstruation, the sloughing off of the uterine lining, days one through five. Days five through fourteen are known as the uterine, proliferate or pre-ovulatory phase. This is earmarked by the thickening of the uterine lining which becomes blood engorge. During the pre-ovulatory phase, estrogen, released by the developing follicle, influences the thickening of the uterus lining. Although estrogen contributes to endometrial thickening, progesterone is primarily responsible for the endometrial thickening. The ovulatory phase overlaps slightly with the pre-ovulatory phase. This normally occurs on day thirteen of the cycle. During ovulation the dominant follicle gets released from the ovary. Estrogen is secreted within the developing follicle. Under the influence of estrogen the follicle continues to mature as the endometrial lining thickens. Luteal cells begin to form within the corpus luteum which serve to secrete estrogen and progesterone this encourages the endometrial lining to be edematous and engorged. This stage is also known as the late luteal phase. The late luteal phase occurs simultaneous to the secretory phase in the endometrial cycle. All of this is done in preparation for fertilization and to support fetal development. If fertilization has not occurred on or about the twenty-fourth day of the cycle, the corpus luteum will degenerate and menstruation will commence in approximately four days, day twenty-eight where menses is marked by vaginal bleeding from the sloughing off of the endometrial lining. [3, 4]
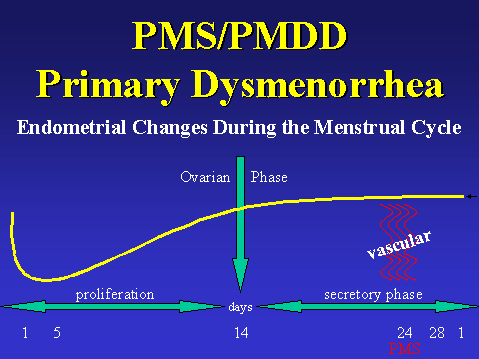
Premenstrual Syndrome, Premenstrual Dysphoric Disorder, and Primary Dysmenorhea:
The cyclic hormones of the female menstrual cycle can augment the possibility for fertilization and nurturing of an embryo to a fetus to a neonate. Menstruation can also create a whirlwind of psycho-emotional, visceral and somatic miseries so much so that it can alter a persons life and affect those around her. In some cases the affects of the pre-menstrual phases of the female reproductive cycle have been used a mitigating tool against manslaughter and even murder. [5] Premenstrual syndrome which occurs during the late luteal phase, is also known as late luteal phase dysphoric disorder. PMS and LLDD consists of a variety of symptoms suffered days prior to menstruation. Though much research exists, the true etiology of PMS and PMDD remains elusive. Most concur that an imbalance of estrogen and progesterone remain as the most probable cause. Other speculated causes include estrogen/progesterone imbalance, nutritional, metabolic/endocrine.
Primary Dysmenorrhea with its’ debilitating pain afflicts women in the pre-menstrual phase. The conundrum is that there are no apparent complaints most common to PMS and PMDD. These women experience unexplained severe pelvic pain absent any pathological disease. These patients must be examined to rule out pelvic diseases, endometriosis, ovarian cysts, polycystic ovary, fibroids, cancers, and pregnancy.
Symptoms of PMS/PMDD and Primary Dysmenorrhea
Symptoms of PMS/PMDD vary from woman to woman and from cycle to cycle. There have been over 150 symptoms identified that contribute to PMS/ PMDD. More than 5 million women in the United States report experiencing pre menstrual like symptoms. [1, 2] According to research reported by Stude approximately 50% of patients in gynecological clinics report the prevalence of PMS. [1]
The most commonly reported symptoms of PMS/LLDD are: bloating, irritability, headaches, migraines, breast tenderness, food cravings, craving sweets or salt, fatigue, feeling of being overwhelmed, abdominal cramping, nausea, back pain and weight gain. The above list is not all inclusive of the myriad symptoms reported by premenstrual women, they are merely a summarization of the most common. Premenstrual hormones create a global disturbance affecting physiology. A commonality is therefore noticed though from cycle to cycle, and from woman to woman, though symptoms experienced can vary greatly and even occur sporadically. It is also common for patients to actually experience these symptoms and not realize that they are pre-menstrual. Women can have perceived weight gain, fatigue, headaches, nausea food cravings and irritability due to infection, thyroid dysfunction or dysglycemia.
Although PMS and PMDD very similar in their presentation there are slight distinctions that separate the diagnosis of each. Those who suffer the symptoms of PMDD describe an increased intensity of symptoms and more emotional conflict. Pelvic pain, common to PMS, PMDD is the earmark symptom for Primary Dysmenorhea. Patients suffering with primary dysmenorrhea report severe pelvic pain during the late luteal phase but decline the other symptoms associated with PMS and PMDD.
Diagnosis of PMS, PMDD and Primary Dysmenorrhea
The criteria for differential diagnosis of PMS, PMDD and Primary Dysmenorrhea are separate and distinct. Separate criteria for each pathology aids in distinguishing one condition from the other. PMS and PMDD are similar except that PMDD is more intense. Patients with Primary Dysmenorrhea are without pelvic pathology. Patients with primary dysmenorrhea emphasize severe cramping.
Diagnostic Parameters for PMS and PMDD
There are diagnostic parameters designed to identify PMS and PMDD. The symptoms must be cyclic, postovulatory, and pre-menstrual. In an updated report written by Dr. Stiener in 1996, Dr. Stiener elaborated on the significant differences between patients suffering from PMS and those who suffer with PMDD. Diagnosis consisting of tracking two cycles via daily charting in conjunction with criteria set forth by the DSM-IV patients could be categorized and offered treatment options accordingly. Essential to diagnosing PMS or PMDD, according to Stiener, would be the inclusion that past cycles must have five or more symptoms that were present for most of the time during the last week of the luteal phase. Symptoms must begin to remit within a few days after the onset of the follicular phase, and were absent in the weeks post menses. A differential factor for diagnosing PMDD is that additionally the patient must experience at least one of the following: [6] marked depressed mood, hopelessness or self depreciating thoughts, marked anxiety, tension, keyed up or on edge, irritability or lability ( sensitive to rejection, sudden sadness or tearfulness), persistent and marked anger, increased interpersonal conflict (6)
Diagnostic Parameters for PMS and PMDD
Other diagnostic criteria for PMS and PMDD includes a symptom free period of at least 7 days during the pre-ovulatory phase, symptoms that must be recurring for at least 3 consecutive cycles, and the absence of a pre-existing psychological condition whereby PMS or PMDD is an exacerbation of such condition. There must be no prior endocrine or metabolic disorders, including abnormal thyroid function, polycystic ovary, mammary dysplasia or galactorrhea. The above conditions must be ruled out for an accurate diagnosis of PMS or PMDD. [1, 2, 4, 6, 7]
Diagnostic Parameters for PMS and PMDD
There is conjecture amongst researchers that the nervous system, especially the sacral plexus, is intricately involved with the symptomotology known as PMS and PMDD. In effectuating an accurate diagnosis, Dr. Stude espoused the effectiveness of using an outcome measured tool, the PMT Cantor (discussed later). In assessing the relationship between the potential for spinal manipulative treatment as a viable treatment option certain criteria must be met. With this in mind Stude clarified the distinctive somatic component purporting in his research that patient must experience at least one somatic and one psychological complaint. [1]
An accurate diagnosis would warrant that a patient experience a symptom free period, that without such would preclude the patient from a diagnosis of PMS or PMDD. Since no biological markers exist to assist in obtaining an accurate diagnosis, diagnostic criteria must be applied to rule out other diseases. It is by this method that PMS or PMDD can be diagnosed.
Dr. Stude discussed the relationship between the female reproductive anatomy and the nerves supplying it. Other authors have identified this relationship and too have espoused the potential in spinal manipulative therapy.
Hormones and Neurotransmitters of PMS and PMDD
It may be refreshing to know many of the psychological and somatic discomforts common to pre-menses has an explanation. Declining levels of seratonin occur in the late luteal phase of the menstrual cycle. This decline often results in mood changes, irritability and depression. Oyelowo reported, in 1997 conducted a qualitative review of relevant literature pertaining to premenstrual syndromes. The review revealed that several of the symptoms of PMS can be explained. Reported was that women with PMS had decreased seretonin level in platelets and whole blood. [4] Progesterone/estrogen imbalance contributes significantly to the retention of water and fluid. It supports feelings of being bloated and can cause a transient weight gain. Excess estrogen has also been linked to nervousness and irritability. Dr. Oyelow also discussed the contribution of decreased dopamine as a contributor. As coenzymes, subclinical levels of both magnesium or of B6 vitamins can greatly obscure the production of dopamine.
Abnormal thyroid function can result in breast tenderness and alterations in normal hormone function. Proper thyroid activity is essential to menstruation and cellular function of the ovaries. Increased activity of the anterior pituitary from hypothyroidism causes the anterior pituitary to release excess amounts of prolactin causing prolactemia. The true effects of hyperprolactemia on PMS and PMDD are inconclusive. Oyelowo reported that her findings indicated that inhibition of prolactin, which rises mid cycle, has a direct affect on breast tenderness during the late luteal phase. [4]
Menstrual Irregularities in Athletic Women:
The Female Athlete Triad
The Female Athlete Triad is a condition consisting of disordered eating, amenorrhea (delayed onset of menses by age 16 years), and premature osteoporosis. Women who engage in excessive exercise or alter their exercise training dramatically and who restrict food consumption are most at risk of compromised health. While the bodies of women who are engaging in extremes of exercise require increased nutrition, those who perform such exercise in pursuit of a slender physique compromise nutritional homeostasis. Rigorous exercise has been positively linked to decreased calcium and decreases in blood estrogen levels. [8–14]
Patients diagnosed with the triad are usually found to have decreased estrogen production, excess cortisol production, and suppressed gonadotrophin-releasing hormone (GnRH). In a qualitative review of select litereature, Morganthal and Resnick reported on research that theorizes that the production of GnRH is inhibited through exercise. The decreased is theorized to be the result of inhibited hypothalamic function, thus interfering with the production of GnRH. [15, 16] Menstrual irregularities, such as amenorrhea, may result from severely reduced caloric intake, coupled with exercise. The combination can result in lack of energy, draining the body and subsequently causing a low T3 thyroid hormone.
Since thyroid function has a direct affect on reproductive function, amenorrhea (defined as 0–3 menstrual cycles per year) can result. [15, 16] Other menstrual irregularities associated with excessive exercise also include delayed menarche, secondary amenorrhea (absence of 3–6 menstrual cycles per year), and oligomenorrhea (cycles longer than 36 days, or four to nine menstrual cycles per year) that can interfere with adequate estrogen production. Hypercortisolemia seen with excess exercise and decreased calorie intake has been positively identified as an impairment to bone density production. It also alters pituitary-hypothalamic function. Concomitant nutritional depletion, hypercortisolemia, and inadequate estrogen production cause premature osteoporosis common to those diagnosed with the female athlete triad. Athletic osteoporosis and resultant stress fractures has been identified as being a direct result of estrogen deficiency from delayed menses and amenorrhea.
Studies have indicated that normal menstruation with cyclic levels of estrogen does not impart alterations in bone accretion. Alterations in cyclic estrogen from excess exercise in fact impairs bone density production. [17, 18] Controversy exists as to whether bone loss is irreversible or can be re-attained. Normal accretion of bone mineral density occurs until the mid-thirties, at which time demineralization may occur. [17] Warren and colleagues report from the literature, that osteopenia in young exercising women with amenorrhea or oligomenorrhea may impair bone formation and suggests a linear relationship between this impairment and estrogen deficiency. Reports from Warren and colleagues and from Drinkwater support the notion that bone accretion is hormonally dependent Estrogen enhances the activity of calcium in forming new bone. The combination of caloric restriction and decreased estrogenemia, in combination can decrease bone density. Results of a questionnaire reported by Thein and Thein revealed that stress fractures occurred at a rate four times greater in women with irregular menses than in those who cycled regularly. (10) Barrow and Saho said long-distance runners with irregular menses were at a significantly greater risk (almost 50%) of developing stress fractures.
Patients should be screened via a patient history, which includes an evaluation of menstrual regularity, prior stress fracture, exercise schedule (number of days/week, duration, and intensity level), and diet. Following positive findings on a history, a bone density test should be ordered to rule out premenopausal osteopenia. For the doctor of chiropractic, this is essential before initiating a care plan that includes osseous adjusting.
Phases of the menstrual cycle: Its’ affect on athletic performance
Increased participation of women in sports has spurred a myriad of studies evaluating the effects of different stages of the menstrual cycle on performance and rate of injury. The three most common stages studied are the premenstrual, menstruation, and intermenstrual. Much controversy exists and reports from these studies remain ambiguous. Moller-Neilson reported on a 1972 study by Wearning demonstrating optimal performance during the inter-menstrual phase, and the poorest at menstruation. Moller-Neilson and Hammar studied female soccer player injuries and the menstrual cycle. They reported the highest occurrence of injuries during the premenstrual and menstrual phases. [19]
Premenstrual or late luteal phase discomforts can cause athletes to lose there competitive edge. Feelings of fatigue, headache, bloating, nausea, back-aches and such can interfere with focus, endurance and strength. Since these symptoms are none to remit several days after the onset of menses, patients retain the risk of injury and impaired performance during that time.
The use of oral contraceptives and their effect on menstrual cycle injury prevention and performance has been studied, but results are inconclusive. Some of the most common side effects of oral contraception include risk of stroke, migraine headaches and weight gain. A list of known side effects can be easily retrieved from the pharmacy. Ask patients to bring the pharmacy print out disclosing the side affects and review it with them. This will make it relatively easy to explain to female patients that oral contraception can actually hinder performance. Managing menstrual function non-invasively, therefore, would appear preferable.
No studies were found evaluating the effect of chiropractic care on the different stages of the menstrual cycle regarding performance or injury. But based on combined information obtained from individual research reported by Korr, Taylor, and Netter, [20–22] the sympathetic nervous system, which innervates the uterus and the blood vessels feeding it, can be regulated by restoration of proper spinal biomechanics. Korr reported on research studies he performed. He identified the causal role between sympathetic nervous system activity and its’ connection to vertebral subluxation. He identified increased sweat gland activity with vertebral malpositioning, whereby normal sweat gland activity was restored with proper restoration of vertebral mobility.
Netter declared that sympathetic innervation to the uterus was a cause for uterine contractile pain. Reports by Taylor indicated that the same sympathetic fibers that innervate the structure, in this case the uterus, also innervates the vasculature that supplies that same structure. Ischemia to that organ/structures can therefore be caused by increased sympathetic activity and its’ affect on blood vessels to that area. Such too can be the cause of pain. This combined information greatly supports the application of chiropractic care as affective in reducing and managing premenstrual uterine cramping.
Any assessment of a physically active female should include a focused gynecological history. The assessment should be obtained at each visit to determine if there is an issue regarding amenorrhea, anorexia, and the premature onset of osteoporosis. Inquiries regarding her age at her first menstrual cycle, frequency of menstruation each year, the duration of her cycle, and whether she bleeds regularly, monthly and on time. If her cycles are heavy, light, or if she just stains. Other inquiries should include having ever been treated for a stress fracture? Ask about her dietary habits. What does she eat for breakfast, lunch, and dinner? Does she indulge in foods she loves? Does she feel guilty? Does she exercise to compensate for this? Does she take a quality multi-vitamin/mineral supplement daily? Developing a questionnaire along these lines will aid in identifying women at risk for the female athlete triad.
Many clinicians see female patients for conditions unrelated to PMS, PMDD or Dysmenorrhea. Inquire about premenstrual type symptoms on the initial visit and in future visits. This can be done in a casual way. The objective is to make your patients aware of there own cycle and any changes since being adjusted. This will help build your practice, as patients will adorn to share this with others suffering from PMS. Additionally your level of awareness will automatically fuel your enthusiasm to discuss the benefits of chiropractic for premenstrual discomforts.
Nutrition
The affects of nutrition regarding maintaining a proper reproductive cycle are profound. Sub- optimal levels of certain nutrients can exacerbate symptoms of PMS, PMDD, and Primary Dysmenorrhea. Vital Nutrients such as: B vitamins, Essential Fatty Acids, Iron, and Magnesium help control symptoms associated with the late luteal phase.B Vitamins are important for controlling PMS/PMDD symptoms. Subclinical levels are associated with decrease dopamine, fatigue, interruption of hepatic estrogen elimination, and depression due to decrease seratonin production from lack of B6. B vitamins, as with all vitamins, should be in a complexed form. Before advising patients on a vitamin regimen, a blood chemistry can be ordered( this may need to be done through their primary care physician) to identify any sub-optimal vitamin or mineral levels.
As coenzymes, the B complex vitamins are involved with metabolism of carbohydrates, fats, and proteins. Sub optimal levels will directly affect energy production. B5, known as the stress vitamin, works with other B vitamins in complex to abate feelings of being overwhelmed or irritable. B6 acts as an anti-inflammatory, dopamine co-factor, and as a coenzyme in the production of seratonin.
Magnesium is responsible for the phosphorylization of B vitamins. Subclinical deficiency indirectly affects symptoms of PMS, PMDD, and Dysmenorrhea due to its direct affect of B vitamin bioavailability. Supplementation of a B complex supplement, significantly decreased both the intensity and the duration of menstrual cramps. [23]
Magnesium is responsible in assisting nerve conduction, muscle function and bioavalability of B vitamins. Deficiencies can activate the cascade of inflammation. It acts as inhibitor to pro-inflammatory modulators, PGE2 and PGF2 alpha. Without optimal levels, systemic inflammation and pain can occur.
Essential Fatty Acids represent another nutritional source for managing PMS and PMDD. Inappropriate amounts of EFAs, omega 3 FAs, in conjunction with dietary intake of improper fat (high omega 6 FAs) will influence inflammatory modulators PGE2 alpha and PGF2 alpha to be produced.
PGE2 alpha and PGF2 alpha
The role of prostaglandins is integral in managing the discomforts of PMS, PMDD and Primary Dysmenorrhea. Certain prostaglandins are responsible for inflammation which happens on a systemic level. PGE2 alpha and PGF2 alpha are the precursors to inflammation. Specific foods contribute to their production or submission such as dairy products and animal fats, which greatly increase production of PGE2a and PGF2a. Essential Fatty Acids especially omega 3 fatty acids, act as precursors to prostaglandins that act as inhibitors to inflammation. Cold water fish and flaxseeds contain the greatest amounts of omega 3 Fatty Acids.
In a study by Koshikawa et al, plasma levels of PGE2a and PGF2a were analyzed through three menstrual cycles. Data collected concluded that increases in plasma prostaglandins will result in symptoms leading to dysmenorrhea. (24)
The Role of PGE2 alpha and PGF2 alpha in Dysmenorrhea
The role of PGE2a prostaglandin production has been researched with astounding results. More than twenty years has since past when Wallace declared a causal role between prostaglandins and dysmenorrhea. By the mid 1970’s research clearly linked menstrual cramping to excessive release of prostaglandins, which was published in the Journal of Obstetrics/Gynecology. PGE2a and PGF2a were confirmed as the specific prostaglandins responsible. Prostaglandins, hormone like substances, are ubiquitous being secreted by tissues throughout the body. The far reaching affects of the prostaglandins PGE2a and PGF2a activity includes physiological actions that includes encouraging smooth muscle contraction (uterus); increasing pain; increasing inflammation; increasing fluid retention; vasoconstriction; migraine headache; mood swings and depression.
Modulating dietary intake of foods that promote the production of PG-2 alpha prostaglandins is an essential component in managing symptoms of the late luteal phase of the menstrual cycle. Increasing intake of milk and diary products will directly increase inflammation, bloating and pain associated with PMS, PMDD, and dysmenorrhea. Increasing dietary intake of foods that promote PGE3 (cold water fish and flax seed are the most abundant in omega 3) will aid in regulating PMS/PMDD and Dysmenorrhea.
Blood Sugar Imbalance of Premenstrual syndromes
Food cravings, increased appetite, and craving sweets are common to those women who menstruate. Blood sugar homeostasis is re-balanced during the late luteal phase. Normal baseline glucose levels of 30mg/dl are raised during this time. The new base-line level that is established forces the body to ingest higher levels of glucose. For those women who crave sweets this offers a sound explanation. If she does not consume enough the body resorts to gluconeogenisis and adrenaline is released. With the release of adrenaline anxiety, heart pounding and symptoms of fear may erupt. Headache, fatigue and dizziness may also be experienced.
The Affect of Spinal Manipulative Therapy on PGE2a and PGF2a
The benefit of chiropractic and osteopathic adjustment are supported in literary reviews. Post treatment women have reported less bloating, headaches, cravings, and cramping to name a few. In a randomized study by Kokjohn et al outcome assessment suggested that the clinical efficacy of spinal manipulative therapy as justified in controlling menstrual pain. Authors stated that pain perception as measured by a Visual Analogue Scale, and the level of menstrual distress measured by the Menstrual Distress Questionnaire, are significantly reduced by spinal manipulative therapy. They associated this reduction with the significant reduction in plasma levels of PGF2a prostaglandin. They did note a similar reduction in plasma PGF2a in both treatment groups; however, the pain and menstrual distress reduction reported were approximately twice as great in the SMT group as in the sham group. [25]
In 1992 Kokjohn et al reported clinical evidence supporting the conjecture that axial adjustments proportionately reduced hyperprostoglandemia. The randomized study consisted of forty-five women (average age 30.3 years). The women where divided into a treatment group and a sham group. The treatment group received axial adjustments at the T10 and L5–S1 levels and to the Sacroiliac joint. The sham group received adjustments to the mid-line base of the sacrum.
The study compared perceived back pain and abdominal pain and perceived menstrual pain. Outcome assessments included VAS (visual analog scale) MDQ (menstrual distress questionnaire) and blood chemistry for circulating plasma levels of PGF2a, 15–keto-13, 14 dihydroprostaglandin. The Vas and MDQ where used fifteen minutes prior to adjusting and at sixty minutes post adjustment. Blood sample where drawn at the same intervals and assayed for KDPGF2a. Results of the VAS and MDQ indicated a decrease in perceived pain. Blood plasma levels illustrated mirrored results. They declared increase in prostaglandin production and that its’ release from the endometrial lining contributes to primary dysmenorrhea. Kokjohn et al hypothesized that primary dysmenorrhea resulted from excess production of prostaglandins PGE2a and PGF2a during menses. Their study concluded that KDPGF2a were significantly lower after intervention. As a caveat to the results, the authors also included that this phenomenon occurred in both the treatment group and the sham group, regardless of the treatment applied. [25] Advise patients to reduce dietary intake of foods that are a pre-cursor to Arachidonic acid omega 6FAs and increase intake of omega 3 FAs. Concluded was that spinal manipulative adjustments are a safe non-pharmaceutical alternative.
Measured Outcome Tools Supporting Spinal Manipulative Therapy for PMS, PMDD and Dysmenorhhea
There are numerous anecdotal case studies supporting the efficacy of chiropractic and osteopathic adjustments in managing PMS, PMDD and Dysmenorrhea. In those studies that incorporated the use of measured outcomes, credence was added to the subjective complaints reported by women. One of the measured outcome tools is the PMT Cator Disc. The PMT-Cator is used to monitor and record subjective scores pre and post adjustment during the premenstrual phase. It converts the patient’s subjective complaints to an objective menstrual cycle score. Patients can report up to five symptoms per day for six weeks. Scores are rated from 0–3, three being the most severe. Scores from post menstrual are subtracted from the premenstrual scores. Scores are concealed to avoid prejudice rating. Analysis and comparative evaluations are more easily procured. This also allows for the potential to analyze larger controlled studies. (1) These types of outcome assessments are necessary to chiropractic as they establish the effectiveness of spinal manipulative therapy for amelioration of symptoms.
Walsh et al performed several studies each illustrating a statistical trend that supports the conjecture of spinal manipulative care as a non-invasive health care option for PMS, PMDD and Dysmenorrhea. In his 1999 release he reported results of a clinical trial of 25 participants with PMS evaluated with the Moos PMS questionnaire. The revised Owestry Low-back Pain Disability and the NDI were used to asses low back pain. Additionally, spinal palpation and orthopedic testing was used. Sixteen of the participants received spinal adjustments, the remaining received placebo treatment. Data was analyzed and scored with paired Student tests and Wilcox signed rank tests. The results of the study supported the hypothesis that spinal adjustment helps PMS symptoms. Limitations to the study do exist as there was an improvement to the sham group but not to the extent of that in the treatment group. [26] In a 1994, Walsh reported on a clinical case where the PMT Cator disc was used to measure outcomes. Of the eight subjects studied in the uncontrolled environment, five of the eight women showed a significant reduction in pre-treatment scores. The authors warn that although these results support anecdotal evidence the group was too small to draw global conclusions. [2]
In a case study, Stude implemented the use of the PMT-Cator for measured outcome in assessing pre and post treatment PMS patients. His efforts purport the use of chiropractic as a treatment method to manage PMS symptoms. By using the PMT-Cator, he demonstrated a significant reduction of symptoms over the course of several months. A spinal adjustment was the only form of treatment used. [1] These are only a few of the many studies and case histories illustrating the benefits of chiropractic.
HYPERSYMPATHETICOTONIA: The neurosomatic connection
Theorist have postulated on the neurologic impact regarding somatic dysfunction and the resulting effect on its’ innervated parts. The female reproductive system represents one of the many organ systems identified as potently affected by sympathetic discord. [1, 20, 21, 22] (The uterus is innervated by the sympathetic trunk, specifically L4 L5, spinal nerves of T11 T12 and afferents from the vagina and perineum from pudendal nerves S2 S3 S4.) It has been well documented that somatic dysfunction can result in hypersympathetic activity causing uterine pain. [1, 20, 21, 22] This supports the conjecture that adjustments to the spinal segmental segment can augment the amelioration of symptoms long associated with PMS, PMDD and Dysmenorrhea.
Sympatheticotonia or hyperactivity of the sympathetic nervous system can have a deleterious affect on physiology. Sympathetic innervation is involved with regulation of smooth muscle contraction (uterus), regulation of cardiac muscle contraction, regulation of endocrine glands: sweat glands and glands of the digestive tract. Sympathetic activity also involves endocrine activity, enzymes and other tissues.
In December 1979 Korr published the findings of his study on hyperactive sympathetic nervous system function and its relation to disease. What he reported validated chiropractic spinal adjustments as a truly valuable tool in managing premenstrual discomforts. Korr monitored the activity of sweat glands and cutaneous vasculature as physiological indicators of sympathetic activity. He noted that topographic changes occurred with sympathetic hyperactivity, which was induced by various types of somatic dysfunction and also with visceral pathology. He showed that a pathological process would occur from hypersympathetic activity from the spinal cord. He set out to prove that musculoskeletal dysfunction, restored through spinal manipulation, re-set neurological output. Korr reported postural faults, repetitive microtrauma and direct injury to the musculoskeletal system could result in sustained hyperactive sympathetic nervous system. [21]
Other studies, including the study by Dishman DJ, found that profound transient attenuation of alpha motor neuron excitability occurred and that amelioration of symptoms occurred immediately post-adjustment. [27, 28]
Pelvic Pain
Pelvic pain can be an enigma at times to both the patient and the treating doctor. Once any pathological conditions, such as endometriosis, ovarian cysts, polycystic ovary, carcinoma, and pelvic disease are ruled out, the clinician can look to the anatomy of the female reproductive system and its’ innervation and vasculature for a formidable recourse to care. Chiropractic and osteopathic adjustments can provide tremendous relief for those in this category. Uterine smooth muscle is innervated by the sympathetic trunk and according to Netter, can be the cause of uterine contractile pain. [22] Korrs’ work confirms this. He documented results of a topographical study. The study indicated a hyperactive sympathetic nervous system as a result of subluxation either from posture or direct injury. In his 1979 paper, Korr specifically states that an imbalance in the autonomic nervous system with sympathetic hyperactivity was implicated in a variety of obstetric and gynecological conditions where uterine contractions where disturbed. [21]
Taylor reports on gynecological pain as a result of its’ vasculature. He notes that the innervation to the part is the same innervation the vasculature of that part has. Therefore, if the uterine vasculature is receiving innervation from the SNS then the uterus becomes ischemic with hypersympathetic activity. [20]
Conclusively, spinal malposition, aberrant motion and stressors of the musculoskeletal system generate the catalyst of hyperactive sympathetic activity, uterine pain during the late luteal phase can be exacerbated by spinal dysfunction. Restoration of proper intersegmental function, according to Korr, restores afferent impulses and can re-balance sympathetic activity. With proper articular, interosseous, muscular, fascial and ligamentous adjustments the tissues involved can report in logical proprioceptive pattern. [21] Conversely, secondary musculoskeletal dysfunction resulting from visceropathological conditions, caused referred pain.
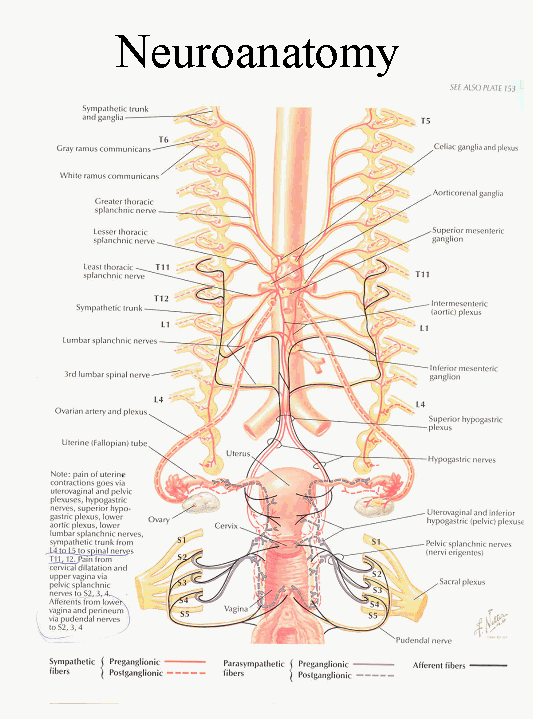
Illustration from Netter identifying the sympathetic
nerves supplying the uterus.
This according to Netter is a source of uterine contractile pain.
References:
Stude D.E.
The management of symptoms associated with premenstrual syndrome
J Manipulative Physiol Ther 1991 (Mar);14 (3):209-216
Walsh et al.
The Efficacy of Chiropractic Therapy on Premenstrual Syndrome: A case Series Study
Chiropractic Journal of Australia Volume 24 Number 4 December 1994: 122-126
Ganong W. M.D.
Review of Medical Physiology , seventeenth edition;
Copyright 1995, Appleton and Lange Company
Oyelowo T.A.
Diagnosis and Management of Premenstrual Syndrome in the Chiropractic Office.
Top Clin Chiro 1997; 4 (3) : 60-67
Kramer LC PMS:
An Old Problem for Today’s Female Worker.
Occup Health Safety 1993; Oct: 49-54
Steiner M.
Premenstrual Dysphoric Disorder: an update
Gen Hosp Psychiatry 1996; 8: 244-50
Brayshaw N. Brayshaw D.
Thyroid Hypofunction in Premenstrual Syndrome.
N Engl. J Med 1986: 315:1486-7
Beim G, Stone D.
Sports medicine issues in the female athlete.
Orthop Clin North Am 1995;26:443-451
Hunter-Griffin LY.
Aspects of injuries to the lower extremity unique to the female athlete.
In: JA Nicholas and EB Hershman, eds.,
The Lower Extremity and Spine in Sports Medicine, 2nd edition.
St. Louis, MO: Mosby, 1995, 141-157
Thein L, Thein J.
The Female Athlete.
J Orthop Sports Phys Ther 1996;23: 134-148
Ardent EA,
Common musculoskeletal injuries in women.
Phys Sports Med 1996;24(7):39-48
Ardent EA,
Orthopaedic issues for active and athletic women.
Phys Sport Med 1994;13:483-503
Baumgardener K.
The female athlete.
Journal of the American Chiropractic Association June 1996;33(6):22-24
Feisler CM.
Special considerations for the female runner.
Journal of Back and Musculoskeletal Rehabilitation 1996;6:37-47
Morgenthal A, Resnick D.
The female athlete: Current concepts.
Topics in Clinical Chiropractic 1997;4(3):11-20
Morgenthal A, Resnick D.
Health-related concerns unique to female athletes.
Topics in Clinical Chiropractic 1997;4(3):51-59
Warren MP, Brooks-Gunn J, Fox RP, Lancelot C, Newman D, Hamilton WG.
Lack of bone accretion and amenorrhea: Evidence for relative osteopenia in weight-bearing bones.
Journal of Clinical Endocrinology and Metabolism 1991;72(4):847-853
Drinkwater BL, Bruemner B, Chestnut CH III.
Menstrual history as a determinant of current bone density in young athletes.
JAMA Jan 26, 1990;263(4):545-548
Moller-Nielsen, J, Hammar, M.
Women’s soccer injuries in relation to the menstrual cycle and oral contraceptive use.
Medicine and Science in Sports and Exercise, 1989.
The American College of Sports Medicine 21(2):126-129 Obstet. Gynecol. 1954 67:1177-96
Taylor HC.
Pelvic Pain Based on a Vascular and Autonomic nervous system disorder.
AmJ Obstet. Gynecol. 1954: 67: 1177-96
Korr IM.
The Spinal Cord as Organizer of Disease Process:
Hyperactivity of Sympathetic Innervation as a Common Factor in Disease.
JAOA 1979; 79 (4) : 232-7
Netter F.,and Colacino S. Ph. D.
Atlas of Human Anatomy, eighth printing;
Cieba-Geigy Corporation, copyright 1989
Wallace J.
Treatment for Painful Menses: A Nutritional and Botanical Approach.
American Journal of Natural Medicine,Vol.. 5, No. December 1998: 21-29
Koshikawa N., Tatsunuma T., Furuya K., Seki K.
Prostaglendins and Premenstrual Syndrome.
Prostaglandin’s Leukotrienes and Essential Fatty Acids (1992) 45, 33-36
Kokjohn K., Schmid D., Triano J. and Brennan P.
The Effect of Spinal Manipulation on Pain and Prostaglandin Levels in Women with Primary Dysmenorrhea.
JMPT; Vol.... 15; Num.. 5; June 1992; 279-285
Walsh BJ, Polus BI.
The Frequency of Positive Common Spinal Clinical Examination Findings in a Sample of
Premenstrual Syndrome Sufferers
Journal of Manipulative and Physiological Therapeutics 1999 (May); 22 (4): 216–220Dishman JD, Bulbulian R.
Spinal Reflex Attenuation Associated with Spinal Manipulation.
Spine 2000 Oct. 1 ; 25 (19): 2519-24; discussion
DishmanJD. And Bulbulian R.
Comparison of Effects of Spinal Manipulation and Massage on Motorneuron Excitability.
Electromyogr. Clin Neurophysiol 2001 Mar; 41 (2): 97-106
Arnould- Frochot S.
Investigation of the Effect of Chiropractic Adjustments on a Specific Gynecological Symptom: Dysmenorrhea
Bulletine, European Cgiropractor’s Union. 1977, 25 (1), pp 17
Walsh M and Polus B. A
Randomized Placebo-Controlled Clinical Trial on the Efficacy of Chiropractic Therapy on Premenstrual Syndrome.
JMPT, Volume 22, Number 9, Novemnber/December 1999; 582-585
Return to WOMEN'S HEALTH
Since 3-28-2005


| Home Page | Visit Our Sponsors | Become a Sponsor |
Please read our DISCLAIMER |