

Characteristics of Paraspinal Muscle Spindle
Response to Mechanically Assisted Spinal
Manipulation: A Preliminary ReportThis section was compiled by Frank M. Painter, D.C.
Send all comments or additions to: Frankp@chiro.org




FROM: J Manipulative Physiol Ther. 2017 (Jul); 40 (6): 371–380 ~ FULL TEXT
OPEN ACCESS William R. Reed, DC, PhD, Joel G. Pickar, DC, PhD,
Randall S. Sozio, BS, LATG, Michael A.K. Liebschner, PhD,
Joshua W. Little, DC, PhD, Maruti R. Gudavalli, PhD
Palmer Center for Chiropractic Research,
Palmer College of Chiropractic,
Davenport, IA.OBJECTIVES: The purpose of this preliminary study is to determine muscle spindle response characteristics related to the use of 2 solenoid powered clinical mechanically assisted manipulation (MAM) devices.
METHODS: L6 muscle spindle afferents with receptive fields in paraspinal muscles were isolated in 6 cats. Neural recordings were made during L7 MAM thrusts using the Activator V (Activator Methods Int. Ltd., Phoenix, AZ) and/or Pulstar (Sense Technology Inc., Pittsburgh, PA) devices at their 3 lowest force settings. Mechanically assisted manipulation response measures included(a) the time required post-thrust until the first action potential,
(b) differences in mean frequency (MF) and mean instantaneous frequency (MIF) 2 seconds before and after MAM, and
(c) the time required for muscle spindle discharge (MF and MIF) to return to 95% of baseline after MAM.RESULTS: Depending on device setting, between 44% to 80% (Pulstar) and 11% to 63% (Activator V) of spindle afferents required >6 seconds to return to within 95% of baseline MF values; whereas 66% to 89% (Pulstar) and 75% to 100% (Activator V) of spindle responses returned to within 95% of baseline MIF in <6 seconds after MAM. Nonparametric comparisons between the 22 N and 44 N settings of the Pulstar yielded significant differences for the time required to return to baseline MF and MIF.
CONCLUSION: Short duration (<10 ms) MAM thrusts decrease muscle spindle discharge with a majority of afferents requiring prolonged periods (>6 seconds) to return to baseline MF activity. Physiological consequences and clinical relevance of described MAM mechanoreceptor responses will require additional investigation.
From the Full-Text Article:
Introduction
Low back pain (LBP) continues to be a major societal health problem, and prevalence rates are only expected to increase with the aging of the American population. [1, 2] More than 80% of the population will experience an episode of LBP during their lives, with 20% to 40% experiencing a reoccurrence of an LBP episode within 1 year after the initial onset in working populations. [3, 4] Low back pain can be treated with multiple approaches including nonpharmacologic, pharmacologic, and surgical interventions. The efficacy, favorable side effect profile, cost effectiveness, and high patient satisfaction associated with the nonpharmacologic approach of spinal manipulation have led to multiple clinical practice guidelines and evidence reports recommending spinal manipulation for certain types of acute and chronic LBP. [5–11] However, the appropriate use and clinical efficacy of spinal manipulation could be greatly enhanced if underlying peripheral and central neurophysiological mechanisms were clearly established. Accomplishing these objectives will require combined contributions from both basic and clinical investigations.
Spinal manipulation traditionally involves applying a single short-lever, high-velocity, low-amplitude (HVLA) thrust of short duration to a targeted dysfunctional vertebral motion segment and/or joint. High-velocity, low-amplitude thrusts can be delivered manually with direct hand contact (≤150 ms thrust duration) or using commercially available mechanically assisted devices (≤10 ms thrust duration). [12–18] Peak forces achieved during a manipulative thrust can vary dramatically depending on anatomic location of treatment, localized tissue compliance, clinician and/or patient’s physical traits, spinal manipulative technique, mechanically assisted device used, and/or device setting. [15–25] A distinct advantage mechanically assisted manipulation (MAM) offers over manual spinal manipulation is that thrust velocity and thrust magnitude can be standardized. Mechanically assisted manipulation is thought to be an effective alternative to manual thrust manipulation based on both comparable measures of vertebrae movements [12, 26–30] and similar clinical outcomes reported in a few smaller studies. [31–34] A recent randomized clinical trial comparing manually delivered spinal manipulation and MAM indicated differences in short-term clinical efficacy, [35] thereby suggesting that distinct physiological mechanisms of action might be responsible for the underlying therapeutic effects of these 2 types of HVLA spinal manipulation.
Proposed physiological mechanisms of action by which spinal manipulation of any type ameliorates musculoskeletal pain include stretch reflex inhibition generated by stretch-induced stimulation of mechanoreceptors or nociceptors in the facet joint capsule and/or the surrounding spinal tissues (muscles, ligaments, discs). [36–43] In humans, the Hoffmann reflex and stretch reflexes have been used to provide a neurophysiological index of α-motoneuron excitability. A commonality shared by a majority of spinal manipulative studies that investigated Hoffmann reflex and stretch reflex responses is the concept that manual therapies mechanistically act by reflexively reducing central α-motoneuron activity via modulating the sensitivity of muscle spindle afferents to muscle stretch. [37, 41–48] Using a computer-controlled feedback motor, we have previously investigated the relationship between various biomechanical parameters of spinal manipulation (thrust duration, thrust magnitude, thrust rate, thrust contact site, etc.) and paraspinal muscle spindle responsiveness in both laminectomy-only and acute facet joint fixation animal models. [49–55] Time to peak impulse in the majority of these animal studies ranged from 75 to 150 ms, which is more closely aligned with manually delivered spinal manipulation compared with the faster time to peak impulse of 2 to 7 ms reported among MAM devices. [15, 18] This extremely short time to peak impulse with MAM devices may result in unique muscle spindle responses and/or central reflex–related mechanisms because these particular mechanoreceptors are sensitive to the rate of muscle stretch.
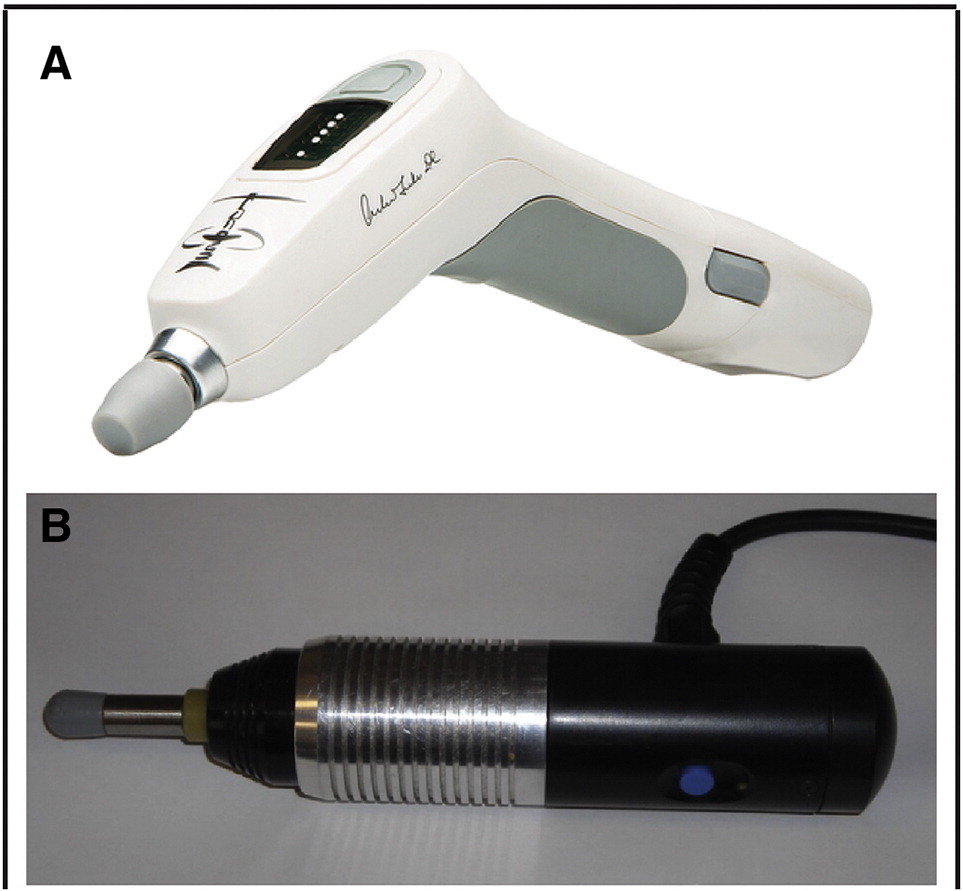
To begin to address the neurophysiological impact of these extremely short MAM thrust durations, we recently conducted a pilot study in a single feline preparation to determine the feasibility of recording muscle spindle afferent responses using an instrumented spring-loaded hammer MAM device (Activator IV; Activator Methods Int. Ltd., Phoenix, AZ). [56] Despite recording from a limited number of muscle spindle afferents, this pilot study clearly indicated that a diverse set of muscle spindle afferent responses are elicited using clinical MAM devices. Post-thrust MAM neural responses ranged from a slight diminution of muscle afferent discharge compared with prethrust to a complete cessation of afferent discharge for nearly 3 seconds. [56] The purpose of this preliminary study is to begin to characterize the temporal effects of 2 electromagnetic solenoid-powered MAM devices (Activator Methods Int. Ltd., Phoenix, AZ [Fig 1A] and Pulstar; Sense Technology Inc., Pittsburgh, PA [Fig 1B]) on muscle spindle discharge to inform future studies.
Figure 1. Photographs of (A) the Activator V, and
(B) Pulstar mechanically assisted manipulation devices.
Discussion
Basic investigations related to neurophysiological changes associated with MAM are important in order to establish underlying mechanisms responsible for spinal manipulation efficacy and to optimize treatment regimens for the 40% to 60% of chiropractic and other manual therapy clinicians in the United States, Canada, Britain, Belgium, New Zealand, and Australia who use these mechanical manipulation devices in some capacity of patient care. [59–64] Despite recent strides being made in determining both the mechanical characteristics and physiological effects of MAM, [13, 15, 17, 18, 25, 56, 65–74] to our knowledge this study is the first to record muscle spindle response evoked using solenoid-powered handheld clinical MAM device technology and only the second to investigate muscle spindle response using any type of MAM device.
This study confirms findings of a previous report using a spring-loaded MAM device (Activator IV) reporting that a range of muscle spindle responses are elicited by MAM. [56] The combined tissue preload and extremely short MAM thrust durations elicited high-frequency discharge from paraspinal muscle spindle afferents followed by a decrease in post-MAM activity. The progressive increase in applied forces typically increased the time required until the first action potential (Table 1 & 3). However, this relationship between applied force and delayed neural response did not necessarily prove to be the case with regard to the amount of time required to return to 95% of baseline MF or MIF. Nonparametric analyses of afferent response to Pulstar thrusts indicated significant differences between 22 N and 44 N thrust settings, with greater time being required for MF/MIF to return to 95% baseline levels for the 44 N MAM thrust but not between the 44 N and 67 N thrust settings. There appeared to be distinct subpopulations of afferent responses, with some returning to baseline activity levels relatively rapidly (<4 seconds), whereas others required much longer periods (>10 seconds) (Tables 2 & 4). It is not known whether these differences are due to inherit differences related to muscle spindle intrafusal fibers types such as bag1, bag2, and/or chain fibers (see Reed et al [75] for greater detail) or to other biological factors.
In the present study we provide post-MAM data regarding changes in both mean frequency (MF) and mean instantaneous frequency (MIF) (Table 1–4), whereas in the past [51, 54] we have primarily focused our attention on MIF changes during the thrust phase (time to peak force) of the spinal manipulation. Because of the lack of tissue preload standardization and handheld vs computer-controlled feedback motor clinical delivery methods, we have chosen to focus on the post-thrust MAM muscle spindle response instead of the spindle response elicited during the thrust. Because muscle spindle afferents typically discharge tonically while at rest, we thought providing the time required to return to 95% baseline activity for both MF and MIF might provide a more complete picture of post-thrust MAM response. As clearly illustrated in Figure 2, MF and MIF recovery do not always occur within close proximity, but generally, these 2 response measures typically occurred within 0.5 to 2 seconds of each other. Comparisons for the Pulstar device indicated significant increases for both MF and MIF when comparing time to return with baseline spindle activity after MAM thrusts delivered at the 22 N and 44 N device settings. We are uncertain how this prolonged delay in return to baseline discharge after MAM affects central interneuron and γ- and α-motoneuron activity, and whether or not these delay differences contribute to the physiological mechanisms or clinical efficacy of spinal manipulation. Answers to these questions will require additional basic and clinical investigation using MAM devices.
Limitations
Preload forces and preload durations were not standardized and MAM devices were not rigidly secured or instrumented to record the actual forces delivered. Instead, MAM devices were handheld as typically used in clinical settings and care was taken to apply the minimum preload necessary to trigger the respective MAM device. Although we tested the 3 lowest settings on each clinical device (with peak forces as low as 22 N), peak forces were not scaled to feline bodyweight per se. Although aspects of a scaling requirement have validity, determining the best scaling parameters is far from clear. Localized thrust forces are quickly dampened by surrounding viscoelastic in vivo tissues, and biological cell response does not scale with body weight. [18, 76] Compared with direct-load cell MAM testing, recent evidence using spinal tissue analogs indicate that applied MAM peak forces are substantially lower (≤59%) based on localized viscoelastic tissue compliance combined with additional compliance from the hand/wrist/forearm when devices are handheld. [18] It should be noted that L7 MAM thrust procedures were not intense enough to tear nearby L6 finely teased afferent fibers from the recording electrode and no tissue damage or spinal fractures occurred with multiple MAM thrusts. Although future studies should consider scaling of applied MAM peak forces, this study highlights the need to determine the actual forces being transmitted through the spine and surrounding tissues in vivo and suggests that these applied forces are likely substantially lower than those reported in the literature after testing on highly idealized beam structures. [18]
Conclusion
This study marks the first time that paraspinal muscle spindle response characteristics related to the delivery of extremely short duration (<10 ms) solenoid-powered MAM thrusts has been reported. Overall, MAM thrusts resulted in decreased muscle spindle discharge with a majority of afferents requiring prolonged periods (>6 seconds) to return to prethrust levels of MF/MIF activity. This neurophysiological effect of MAM far outlasts the 2– to 7–ms stimulus duration, but determination as to how this prolonged delay in return to baseline activity levels affects spinal or supraspinal mechanosensory processing or clinical outcomes will require additional investigation.
Practical Applications
This study found that short duration (<10 ms) mechanically assisted
lumbar manipulative thrusts decreases muscle spindle discharge
relative to baseline activity.It required prolonged periods (>6 seconds) for paraspinal muscle spindles
to return to their prethrust resting discharge activity levels after short
duration (<10 ms) mechanically assisted spinal manipulative thrusts.This study suggests that subpopulations of muscle spindle afferents respond
differently to short duration mechanically assisted spinal manipulative thrusts.These findings may become important to patient care if future studies report
that mechanosensory response is a mechanism that contributes to
the clinical efficacy of spinal manipulation.
Return to INSTRUMENT ADJUSTING
Since 6–22–2017


| Home Page | Visit Our Sponsors | Become a Sponsor |
Please read our DISCLAIMER |